Comprehensive Analysis of Lithium Plating in Lithium-Ion Batteries: From Principles to Prevention Lithium - Ion Battery Equipment
Comprehensive Analysis of Lithium Plating in Lithium-Ion Batteries: From Principles to Prevention Lithium - Ion Battery Equipment
Lithium-ion batteries, as the most prevalent electrochemical energy storage devices today, are widely used in smartphones, laptops, electric vehicles, and other applications. However, during operation, a phenomenon known as lithium plating can severely impact battery performance and safety. This article systematically explores the mechanisms, hazards, and preventive measures for lithium plating, providing a thorough understanding of this critical issue affecting lithium-ion battery performance. From the fundamental definition of electrode potential to complex thermodynamic analysis; from microscopic observations in laboratories to macroscopic manifestations in real-world applications; from material design optimizations to intelligent battery management systems—we delve layer by layer into the scientific principles behind lithium plating and discuss effective strategies for its mitigation in practical scenarios.
Electrode Potential and the Thermodynamic Basis of Lithium Plating
In lithium-ion batteries, electrode potential is a core concept that directly determines the direction and extent of chemical reactions. To understand lithium plating, we must first clarify the definition of electrode potential. The potential is typically referenced to the Li/Li⁺ electrode, analogous to using sea level as the zero point for altitude. When lithium metal is immersed in an electrolyte containing Li⁺ ions, a stable potential is established at its surface, defined as 0 V (vs. Li/Li⁺). At this reference point, the redox equilibrium between metallic lithium (Li⁰) and lithium ions (Li⁺) is maintained.
Potential levels directly reflect the energy state of electrons. In electrochemical systems, a lower (more negative) electrode potential indicates higher-energy electrons, which are more likely to participate in reduction reactions. During charging, the lithiation reaction at the anode is essentially a reduction process: Li⁺ + e⁻ + C₆ → LiC₆. Whether this reaction proceeds spontaneously depends on the Gibbs free energy change (ΔG). According to electrochemical thermodynamics, ΔG = -nFE, where n is the number of transferred electrons, F is Faraday’s constant, and E is the deviation from the equilibrium potential.
When the anode potential is above 0 V (vs. Li/Li⁺), the electron energy is insufficient to reduce Li⁺ to metallic lithium but sufficient to drive Li⁺ intercalation into graphite layers. This is because graphite provides a stabilizing effect, with an intercalation equilibrium potential (~0.1–0.2 V) slightly higher than lithium deposition (0 V). Here, ΔG_embed < 0 (spontaneous intercalation), while ΔG_deposit > 0 (deposition is thermodynamically forbidden).
However, when the anode potential drops below 0 V, the situation changes fundamentally. The high-energy electrons can now reduce Li⁺ to metallic lithium. Thermodynamically, when E < 0 V, ΔG_deposit < 0, making lithium plating spontaneous. In practical batteries, once graphite intercalation sites near saturation, continued charging drives the potential below 0 V, triggering plating.
The Nernst equation further explains this relationship. The equilibrium potential for lithium deposition (E_eq,deposit) is 0 V, while the actual potential E = E_eq,deposit - (RT/nF)ln(Q). When Li⁺ concentration rises or available sites diminish, Q changes, lowering E. Once E < E_eq,deposit, the system deposits lithium to restore equilibrium.
This thermodynamic principle is crucial for battery design. The same logic applies to sodium-ion batteries—when the anode potential falls below sodium’s deposition potential (vs. Na/Na⁺), sodium plating occurs. Thus, different battery systems share a common thermodynamic origin for metal plating, albeit with varying threshold potentials.
Microscopic Mechanisms and Kinetic Processes of Lithium Plating
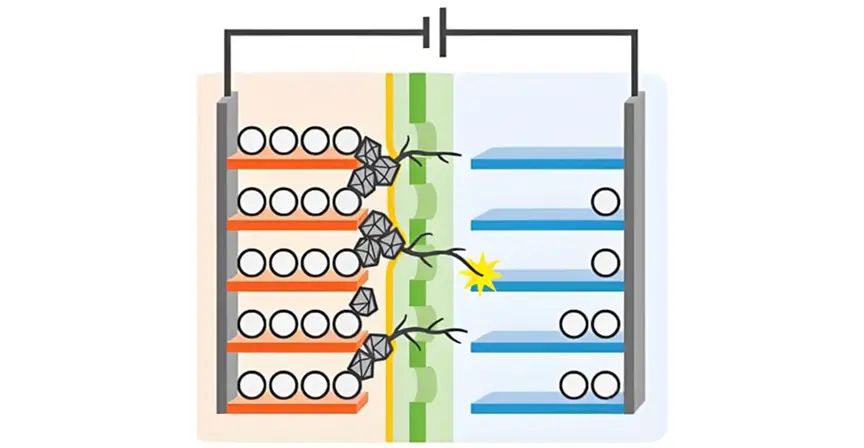
Lithium plating is far from a simple thermodynamic equilibrium issue; it involves intricate microscopic mechanisms and kinetic processes. At the microscopic level, plating occurs when Li⁺ ions gain electrons at the anode surface, transitioning from ionic to metallic states. Normally, Li⁺ intercalates into graphite (forming LiC₆) at 0.1–0.2 V (vs. Li/Li⁺). However, when kinetic balance is disrupted, this process deviates.
The space charge layer theory explains plating initiation. As Li⁺ migrates from the electrolyte to the anode, a charge-rich zone forms at the interface. If ion migration lags behind electron transfer, Li⁺ accumulates locally. When surface concentration exceeds ~1.2 mol/m³, the anode potential drops below 0 V, inducing plating.
Plating unfolds in two stages:
- Nucleation: Li⁺ gains electrons at surface defects, forming metallic nuclei. This stage faces an energy barrier, causing potential fluctuations.
- Growth: Post-nucleation, lithium grows on existing nuclei, stabilizing the potential. Scanning electron microscopy reveals initial granular deposits evolving into mossy or dendritic structures.
Dendrite morphology is influenced by:
- Current density: High currents promote needle-like dendrites.
- Temperature: Low temperatures yield finer, denser dendrites.
- Electrolyte additives: Some additives encourage bulkier deposits.
Kinetically, plating results from charge transfer limitation (CTL) and solid diffusion limitation (SDL). As charging progresses, graphite intercalation sites deplete, slowing Li⁺ diffusion, while electrolyte Li⁺ continues migrating to the anode. This imbalance forces the potential toward plating levels.
Temperature significantly impacts kinetics. Below 0°C, electrolyte viscosity rises, ion conductivity drops, and graphite’s intercalation activation energy increases. Studies show -20°C fast-charging raises plating risk fivefold versus 25°C.
Interface properties are equally critical. The solid electrolyte interphase (SEI) governs Li⁺ penetration. An ideal SEI has uniform ion channels and moderate thickness. Excessive or uneven SEI impedes intercalation, while SEI defects cause localized plating. Moisture (>500 ppm) or improper formation cycles degrade SEI, elevating plating risks.
At the atomic scale, lithium’s crystal orientation affects dendrite shape. Li prefers (110) plane growth, forming high-aspect-ratio dendrites. Additives like Cs⁺ or Rb⁺ can alter growth directions via electrostatic shielding, yielding denser deposits.
Primary Causes and Manifestations of Lithium Plating
Lithium plating stems from multiple synergistic factors, broadly categorized into:
- Insufficient anode margin (N/P ratio < 1):
- Leads to uniform plating, "yin-yang" plating (one-sided), or stripe-like plating from uneven Coating.
- Improper charging protocols:
- Low-temperature charging: Slows Li⁺ diffusion, causing surface accumulation.
- High-rate charging: At >1C, intercalation can’t match charging speed, dropping effective N/P ratio to 0.5.
- Overcharging: Forces Li⁺ reduction when graphite is saturated.
- Blocked intercalation paths:
- Wrinkled separators or cell deformation create local dead zones with adjacent plating.
- Material defects:
- Excessive anode compaction (>1.7 g/cm³), poor electrolyte wetting (<48h aging), or SEI defects.
- Special positional flaws:
- Overhang regions (>2 mm) or edge-thickening during coating cause edge plating.
Plating morphology reveals root causes:
- Edge plating: Overhang or coating defects.
- Localized plating: Random spots from material flaws or pressure points.
- Uniform plating: Whole-anode coverage from overcharging or low N/P ratio.
Advanced detection methods like ultrasonic scanning (10 μm resolution) and infrared imaging enable real-time monitoring.
Multifaceted Hazards of Lithium Plating
Plating inflicts irreversible damage across multiple fronts:
- Capacity fade: Plated lithium reacts with electrolytes, forming inert compounds (e.g., Li₂CO₃), permanently depleting active lithium. Plated cells may retain only 75% capacity after 50 cycles (vs. 92% for normal cells).
- Safety crises: Dendrites pierce separators, causing short circuits, thermal runaway, fires, or explosions.
- Impedance surge: Dendrites rupture SEI, increasing ion transport resistance and heat generation.
- Gas generation: Reactions like Li + DMC → LiOCOOCH₃ + ½H₂↑ swell cells, risking casing rupture.
- Nonlinear lifespan impact: Initial plating may temporarily boost capacity (reversible Li contribution), but beyond ~1% total lithium, performance collapses.
Microstructurally, plating shatters graphite, fracturing particles and conductive networks, accelerating degradation.
Systemic Solutions to Lithium Plating
A multi-layered defense integrates innovations across the battery lifecycle:
- Material advances:
- Silicon anodes: 10× higher capacity than graphite, though volume expansion requires nanoengineering.
- Hard carbon: Nano-pores enhance low-temperature performance.
- Pre-lithiation: Compensates initial Li loss.
- Electrolyte optimization:
- Low-viscosity solvents (FEC, EMC) and SEI-forming additives (VC, PS).
- Solid-state electrolytes physically block dendrites.
- Design & manufacturing precision:
- N/P ratio ≥1.1, anode loading <12 mg/cm², porosity 25–35%, overhang <2 mm.
- Dry electrode tech (e.g., Tesla’s 4680 cells) ensures uniformity.
- Smart battery management:
- Low-temperature current limiting (<0.5C at <10°C).
- Multi-stage charging (CC-CV-pulse).
- SOC limits (80–95% daily, 100% pre-travel).
- AI-driven real-time adjustments.
- Thermal management:
- Pre-heating to >15°C in cold climates.
- Liquid cooling maintains ≤2°C cell-to-cell variation.
Industry leaders exemplify success:
- CATL: "Self-healing SEI" Cuts -20°C plating risk by 90%.
- Tesla: 4680’s tabless design reduces polarization.
- BYD: Blade battery’s thin design enhances heat dissipation.
Future directions include:
- 3D current collectors for uniform Li deposition.
- AI-optimized charging curves.
- Self-repairing additives (e.g., LiNO₃).
A full-chain防控体系—spanning materials, design, manufacturing, and management—is essential to balance high energy density, fast charging, and wide-temperature operation with safety and longevity, propelling sustainable energy storage forward.










